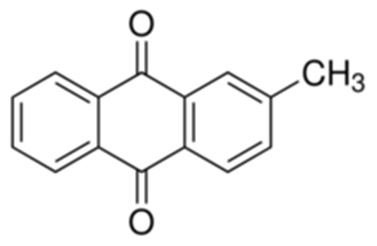
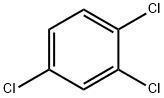
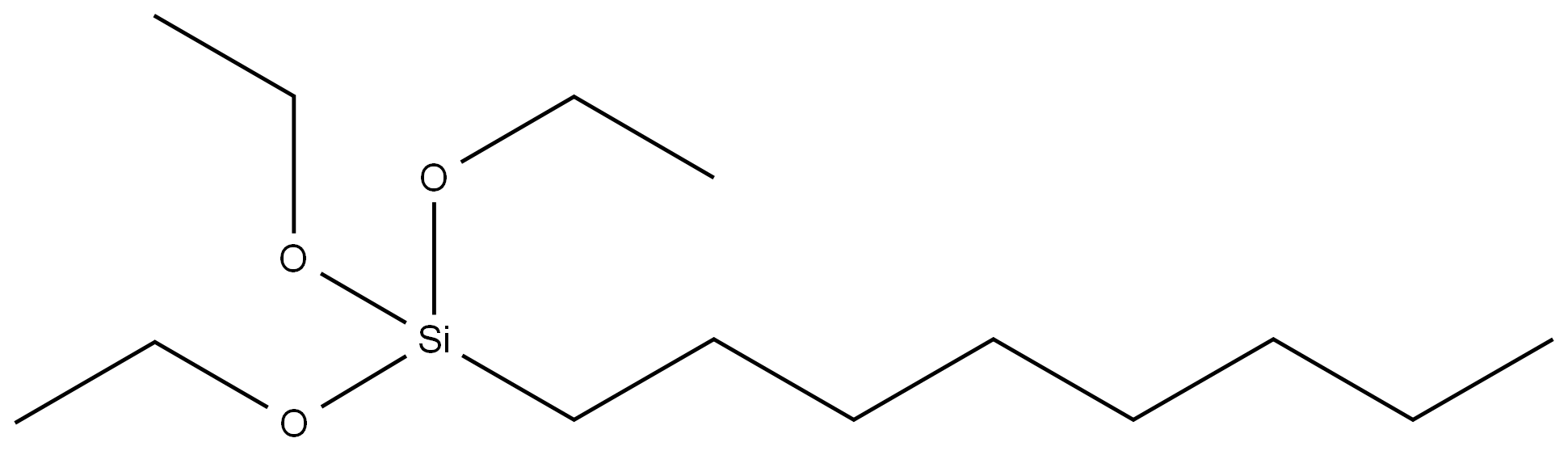
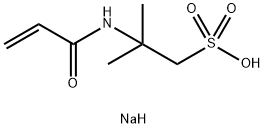
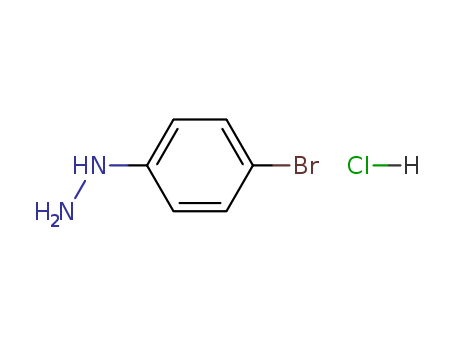4-BROMOPHENYLHYDRAZINE HYDROCHLORIDE literature
Rhodium(III)-catalyzed in situ oxidizing directing group- assisted c-h bond activation and olefination: A route to 2-vinylanilines
Muralirajan, Krishnamoorthy,Haridharan, Radhakrishnan,Prakash, Sekar,Cheng, Chien-Hong
, p. 761 - 766 (2015)
A new and efficient method for the synthesis of 2-vinylanilines from the reaction of arylhydrazine hydrochlorides with alkenes and diethyl ketone via a rhodium-catalyzed C-H activation is described. The oxidant-free olefination reaction involves the in situ generation of an -N-N=CR1R2 moiety as the oxidizing directing group thus providing an easy access to 2-vinylanilines.
Design, Synthesis, and Antifungal Activity of 2,6-Dimethyl-4-aminopyrimidine Hydrazones as PDHc-E1 Inhibitors with a Novel Binding Mode
Zhou, Yuan,Zhang, Shasha,Cai, Meng,Wang, Kaixing,Feng, Jiangtao,Xie, Dan,Feng, Lingling,Peng, Hao,He, Hongwu
, p. 5804 - 5817 (2021/06/25)
A series of novel 2,6-dimethyl-4-aminopyrimidine hydrazones 5 were rationally designed and synthesized as pyruvate dehydrogenase complex E1 (PDHc-E1) inhibitors. Compounds 5 strongly inhibited Escherichia coli (E. coli) PDHc-E1 (IC50 values 0.94-15.80 μM). As revealed by molecular docking, site-directed mutagenesis, enzymatic, and inhibition kinetic analyses, compounds 5 competitively inhibited PDHc-E1 and bound in a "straight"pattern at the E. coli PDHc-E1 active site, which is a new binding mode. In in vitro antifungal assays, most compounds 5 at 50 μg/mL showed more than 80% inhibition against the mycelial growth of six tested phytopathogenic fungi, including Botrytis cinerea, Monilia fructigena, Colletotrichum gloeosporioides, andBotryosphaeria dothidea. Notably, 5f and 5i were 1.8-380 fold more potent against M. fructigena than the commercial fungicides captan and chlorothalonil. In vivo, 5f and 5i controlled the growth of M. fructigena comparably to the commercial fungicide tebuconazole. Thus, 5f and 5i have potential commercial value for the control of peach brown rot caused by M. fructigena.
Highly Efficient Synthesis of Hindered 3-Azoindoles via Metal-Free C-H Functionalization of Indoles
Guillemard, Lucas,Jacob, Nicolas,Wencel-Delord, Joanna
supporting information, p. 574 - 580 (2020/02/13)
Although 3-azoindoles have recently emerged as an appealing family of photoswitch molecules, the synthesis of such compounds has been poorly covered in the literature. Herein a high-yielding and operationally simple protocol is reported allowing the synthesis of 3-azoindoles, featuring important steric hindrance around the azo motif. Remarkably, this C-H coupling is characterized by excellent atom economy and occurs under metal-free conditions, at room temperature, and within few minutes, delivering the expected products in excellent yields (quantitatively in most of the cases). Accordingly, a library of new molecules, with potential applications as photochromic compounds, is prepared.
Heterocyclic com pounds and organic light-emitting diode including the same
-
Paragraph 0657-0663, (2021/01/19)
The present invention relates to a novel heterocyclic compound and an organic electroluminescent device comprising the same. The heterocyclic compound is represented by the following Chemical Formula 1, and the organic electroluminescent device including the heterocyclic compound has excellent driving voltage, luminous efficiency, and lifespan properties. Chemical Formula 1. (by machine translation)
An electroluminescen compound and an electroluminescent device comprising the same
-
Paragraph 0390-0394, (2021/02/02)
The present invention relates to an organic light-emitting compound represented by chemical formula 1. An organic electroluminescent device comprising the organic light-emitting compound in the present invention has excellent power efficiency, light-emitting efficiency, and long life cycle because the present invention can be operated by a lower driving-voltage in comparison with a device comprising conventional phosphorescent host materials.