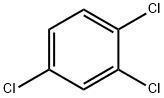
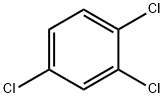
1,2,4-Trichlorobenzene literature
Catalytic Dehalogenation of Highly Chlorinated Benzenes and Aroclors Using PdCl2(dppf) and NaBH4: Efficiency, Selectivity, and Base Support
Lassová, Luisa,Lee, Hian Kee,Hor, T.S. Andy
, p. 3538 - 3543 (1998)
Reported herein is a convenient one-pot system that can dehalogenate highly chlorinated benzenes at room temperature with reasonable conversion rates using PdCl2(dppf) (dppf = 1,1′-bis-(diphenylphosphino)ferrocene) as catalyst, NaBH4 as reducing agent, TMEDA (N,N,N',N'-tetra-methyl-1,2-ethylenediamine) as supporting base, and THF as solvent. Total conversion of substrate to less chlorinated isomers is achieved within 200 h when hexachloro-, pentachloro-, and tetrachlorobenzenes are used. Degradation to benzene is not achievable, but the efficiency shown in the partial dechlorination is encouraging. A pronounced selectivity is accomplished with removal of meta-substituted chlorines being preferred over ortho- or para-substituted Cl atoms. The sequence in which reagents are added is also critical, thus indicating a protective role of the base. The effectiveness of the method was tested on the PCB mixtures Aroclor 1242, 1248, and 1254. Dechlorination efficiency at 67 °C is satisfactory.
Antagonism/agonism modulation to build novel antihypertensives selectively triggering I1-imidazoline receptor activation
Del Bello, Fabio,Bargelli, Valentina,Cifani, Carlo,Gratteri, Paola,Bazzicalupi, Carla,Diamanti, Eleonora,Giannella, Mario,Mammoli, Valerio,Matucci, Rosanna,Micioni Di Bonaventura, Maria Vittoria,Piergentili, Alessandro,Quaglia, Wilma,Pigini, Maria
, p. 496 - 501 (2015)
Pharmacological studies have suggested that I1-imidazoline receptors are involved in the regulation of cardiovascular function and that selective I1-agonists, devoid of the side effects associated with the common hypotensive α2<
Photoreductive dehalogenation of halogenated benzene derivatives using ZnS or CdS nanocrystallites as photocatalysts
Yin, Hengbo,Wada, Yuji,Kitamura, Takayuki,Yanagida, Shozo
, p. 227 - 231 (2001)
ZnS nanocrystallites (nc-ZnS) prepared in N,N-dimethylformamide (DMF) photocatalyze dehalogenation of halogenated benzenes to benzene as the final product from chlorinated benzenes and to difluorobenzenes from fluorinated benzenes in the presence of triethylamine (TEA) as an electron donor under UV light irradiation (λ > 300 nm). When CdS nanocrystallites (nc-CdS) are used as a photocatalyst (λ > 400 nm), halogenated benzenes are photoreductively dehalogenated, yielding trichlorobenzene from hexachlorobenzene and tetrafluorobenzene isomers from hexafluorobenzene as the final products. Photoformed electrons on nc-ZnS and nc-CdS have such negative reduction potentials that these electrons reduce polyhalogenated benzenes, leading to the successive dehalogenation. nc-ZnS exhibits higher photocatalytic activity than nc-CdS due to the more negative potential of the electrons on nc-ZnS than that on nc-CdS. The higher activities of nc-ZnS and nc-CdS compared to their bulk forms are explained as being due to their quantum size effects and the adsorptive interaction between the substrates and the nanosized photocatalysts. ZnS nanocrystallites (nc-ZnS) prepared in N,N-dimethylformamide (DMF) photocatalyze dehalogenation of halogenated benzenes to benzene as the final product from chlorinated benzenes and to difluorobenzenes from fluorinated benzenes in the presence of triethylamine (TEA) as an electron donor under UV light irradiation (λ>300 nm). When CdS nanocrystallites (nc-CdS) are used as a photocatalyst (λ>400 nm), halogenated benzenes are photoreductively dehalogenated, yielding trichlorobenzene from hexachlorobenzene and tetrafluorobenzene isomers from hexafluorobenzene as the final products. Photoformed electrons on nc-ZnS and nc-CdS have such negative reduction potentials that these electrons reduce polyhalogenated benzenes, leading to the successive dehalogenation. nc-ZnS exhibits higher photocatalytic activity than nc-CdS due to the more negative potential of the electrons on nc-ZnS than that on nc-CdS. The higher activities of nc-ZnS and nc-CdS compared to their bulk forms are explained as being due to their quantum size effects and the adsorptive interaction between the substrates and the nanosized photocatalysts.
Dehalogenation of o-dihalogen substituted arenes and α,α′-dihalogen substituted o-xylenes with lanthanum metal
Nishiyama, Yutaka,Kawabata, Hiroshi,Nishino, Toshiki,Hashimoto, Kouji,Sonoda, Noboru
, p. 6609 - 6614 (2003)
It was found that lanthanum metal caused the dehalogenation of o-dihalogen substituted arenes and α,α′-dihalogen substituted o-xylenes to generate the corresponding benzynes and o-quinodimethanes. When o-dihalogen substituted arenes were allowed to react with lanthanum metal in the presence of dienes, the Diels-Alder products between benzyne and dienes were formed in moderate to good yields. Similarly, the Diels-Alder adducts of o-quinodimethane with dienophiles were obtained, in the reaction of α,α′-dibromo-o-xylenes with lanthanum metal in the presence of dienophiles.
-
Kauer,DuVall,Alquist
, p. 1335,1336 (1947)
-
Iron(iii)porphyrin electrocatalyzed enantioselective carbon-chloride bond cleavage of hexachlorocyclohexanes (HCHs): Combined experimental investigation and theoretical calculations
Liang, Xu,Li, Minzhi,Mack, John,Lobb, Kevin,Zhu, Weihua
, p. 11470 - 11476 (2018/08/28)
Enantioselective electrocatalysis of α-, β-, γ- and δ-hexachlorocyclohexanes (HCHs) by tetrakis-pentafluorophenyl-Fe(iii)porphyrin is described. The first example of the combined use of electrochemical measurements and theoretical calculations to determine the mechanism of the enantioselective C-Cl bond cleavage of the electrocatalysis is reported. The electrochemical measurements demonstrate that the reactivity of the HCHs follows the order γ-HCH > α-HCH > δ-HCH > β-HCH. Steric considerations and a molecular orbital theory approach can be used to rationalize the enantioselective nature of the catalysis based on the ease of approach of each Cl atom to the central Fe(i) ion and a consideration of the nodes on the C-Cl bonds that weaken these bonds in a manner that results in bond cleavage and the formation of an Fe-Cl bond.
PROCESS FOR THE PREPARATION OF ORGANIC HALIDES
-
Paragraph 00146, (2017/08/01)
The present invention provides a halo-de-carboxylation process for the preparation of organic chlorides, organic bromides and mixtures thereof, from their corresponding carboxylic acids, using a chlorinating agent selected from trichloroisocyanuric acid (TCCA), dichloroisocyanuric acid (DCCA), or combination thereof, and a brominating agent.
Dicamba preparation process
-
Paragraph 0051; 0052; 0053, (2017/04/03)
The invention belongs to the technical field of herbicide dicamba preparation and relates to a dicamba preparation process. The dicamba preparation process includes steps: taking benzene as a raw material to generate 1,2,4-trichlorobenzene through directional chlorination, catalysis, re-chlorination and rectification; hydrolyzing the 1,2,4-trichlorobenzene to generate a mixture of 2,5-dichlorophenol and 2,4-dichlorophenol, and separating and purifying to obtain 2,5-dichlorophenol; using the 2,5-dichlorophenol to prepare 3,6-dichlorosalicylic acid; subjecting the 3,6-dichlorosalicylic acid to methylation, saponification, acidification and the like to obtain dicamba. By optimization of technical steps and parameters, the whole dicamba preparation process has advantages of simplicity, low cost, high yield, high selectivity, remarkable reduction of wastewater and increase of equipment utilization rate.
A method for the synthesis of 2, 5 - dichlorophenol (by machine translation)
-
Paragraph 0064, (2016/10/09)
The invention relates to a 2, 5?Dichiorophenol synthetic method, which belongs to the technical field of the synthesis of the key intermediate chamber. The invention is composed of a pure chlorization generated 1, 2, 4?Trichlorobenzene and santochlor is easy to separate, thereby avoiding the difficulties caused by the separating of the high production cost and low production efficiency; because the 1, 2, 4?Trichlorobenzene and to two chiorophenoxy can be obtained respectively 2, 5?Dichiorophenol, utilization rate of raw materials is high, thereby avoiding the waste of the by-product, so that the production cost is greatly reduced. In addition, the two paradichlorbenzene through two different method to obtain 2, 5?Dichiorophenol. The synthesis technique of this invention is simple, the production cost is low, the utilization rate of raw materials is high, and the craft there are few by-products, less generation of three wastes, is more suitable for large-scale industrial production. (by machine translation)