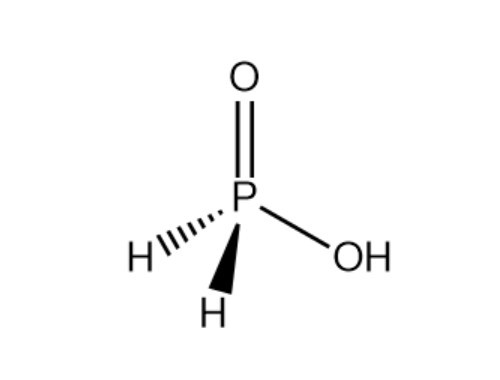
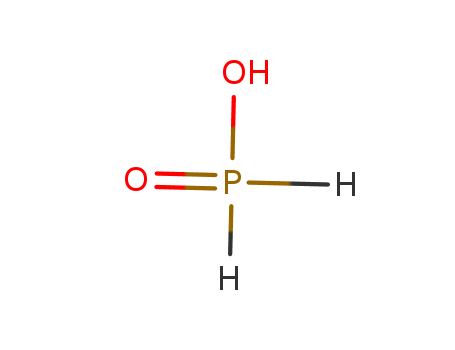
Hypophosphorous acid literature
Synthesis of monophosphines directly from white phosphorus
Scott, Daniel J.,Cammarata, Jose,Schimpf, Maximilian,Wolf, Robert
, p. 458 - 464 (2021/04/09)
Monophosphorus compounds are of enormous industrial importance due to the crucial roles they play in applications such as pharmaceuticals, photoinitiators and ligands for catalysis, among many others. White phosphorus (P4) is the key starting material for the preparation of all such chemicals. However, current production depends on indirect and inefficient, multi-step procedures. Here, we report a simple, effective ‘one-pot’ synthesis of a wide range of organic and inorganic monophosphorus species directly from P4. Reduction of P4 using tri-n-butyltin hydride and subsequent treatment with various electrophiles affords compounds that are of key importance for the chemical industry, and it requires only mild conditions and inexpensive, easily handled reagents. Crucially, we also demonstrate facile and efficient recycling and ultimately catalytic use of the tributyltin reagent, thereby avoiding the formation of substantial Sn-containing waste. Accessible, industrially relevant products include the fumigant PH3, the reducing agent hypophosphorous acid and the flame-retardant precursor tetrakis(hydroxymethyl)phosphonium chloride. [Figure not available: see fulltext.]
Oxidation of white phosphorus by peroxides in water
Abdreimova,Akbaeva,Polimbetova
, p. 1873 - 1876 (2017/09/27)
A mixture of hypophosphorous, phosphorous, and phosphoric acids is formed during the anaerobic oxidation of white phosphorus by peroxides [ROOН; R = Н, 3-ClC6H4CO, (СН3)3С] in water. The rate of reactions grows considerably upon adding nonpolar organic solvents. The activity series of peroxides and solvents are determined experimentally. NMR spectroscopy shows that the main product of the reaction is phosphorous acid, regardless of the nature of the peroxide and solvent. A radical mechanism of oxidation of white phosphorus by peroxides in water is proposed. It is initiated by the homolysis of peroxide with the formation of НO? radicals that are responsible for the homolytic opening of phosphoric tetrahedrons. Further oxidation and stages of the hydrolysis of intermediate phosphorus-containing compounds yield products of the reaction.
Reactivity of phosphine oxide H3PO in the reactions with ketones
Gorbachuk,Badeeva,Babaev,Rizvanov, I. Kh.,Zinnatullin,Pavlov,Khayarov, Kh. R.,Yakhvarov
, p. 1289 - 1294 (2017/02/05)
The reactivity of the electrochemically generated phosphine oxide H3PO towards ketones (acetone, ethyl methyl ketone, methyl n-propyl ketone, and tert-butyl methyl ketone) has been studied. It was found that this reaction led to the formation o
Electrochemical generation of P4 2- dianion from white phosphorus
Yakhvarov,Gorbachuk,Khayarov, Kh. R.,Morozov,Rizvanov, I. Kh.,Sinyashin
, p. 2423 - 2427 (2015/08/03)
Electrochemical reduction of elemental (white) phosphorus in an undivided cell equipped with a sacrificial metal anode (Al, Co, Nb, Sn) results in the formation of the reduced form of white phosphorus, P4 2- dianion, which was detected in solution by 31P NMR spectroscopy.