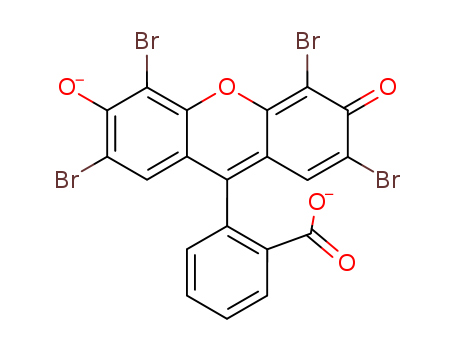
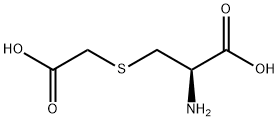
Acid Red 87 Application
Application
It is primarily used in the manufacture of red ink and red pencils; however, it is suitable only for dyeing carpets, as its fastness is poor when applied to other textiles. After purification, Acid Red 87 can be utilized as a coloring agent for pharmaceuticals and cosmetics, as well as for dyeing leather; furthermore, its lead salt serves as a pigment.
Market
United States, Germany, India, Malaysia etc
Acid Red 87 literature
Exponential Amplification Using Photoredox Autocatalysis
Kim, Seunghyeon,Martínez Dibildox, Alejandra,Aguirre-Soto, Alan,Sikes, Hadley D.
, p. 11544 - 11553 (2021)
Exponential molecular amplification such as the polymerase chain reaction is a powerful tool that allows ultrasensitive biodetection. Here, we report a new exponential amplification strategy based on photoredox autocatalysis, where eosin Y, a photocatalyst, amplifies itself by activating a nonfluorescent eosin Y derivative (EYH3-) under green light. The deactivated photocatalyst is stable and rapidly activated under low-intensity light, making the eosin Y amplification suitable for resource-limited settings. Through steady-state kinetic studies and reaction modeling, we found that EYH3- is either oxidized to eosin Y via one-electron oxidation by triplet eosin Y and subsequent 1e-/H+ transfer, or activated by singlet oxygen with the risk of degradation. By reducing the rate of the EYH3- degradation, we successfully improved EYH3-to-eosin Y recovery, achieving efficient autocatalytic eosin Y amplification. Additionally, to demonstrate its flexibility in output signals, we coupled the eosin Y amplification with photoinduced chromogenic polymerization, enabling sensitive visual detection of analytes. Finally, we applied the exponential amplification methods in developing bioassays for detection of biomarkers including SARS-CoV-2 nucleocapsid protein, an antigen used in the diagnosis of COVID-19.
Designing the selectivity of the fluorescent detection of amino acids: A chemosensing ensemble for histidine
Hortala, Marta Ansa,Fabbrizzi, Luigi,Marcotte, Nathalie,Stomeo, Floriana,Taglietti, Angelo
, p. 20 - 21 (2007/10/03)
The imidazole group of histidine deprotonates and bridges the two CuII centers of a dimetallic polyamine macrocyclic complex, displacing the previously bound and quenched fluorescent indicator I. Thus, histidine recognition is signaled by the revival of the fluorescence of I. Selectivity with respect to other natural amino acids is achieved by choosing an indicator of tuned affinity toward the dicopper(II) receptor. Copyright
Metalocarboxypeptidase inhibitors and derived molecules used as antitumor agents
-
, (2008/06/13)
The present invention relates to metalocarboxypeptidase inhibitors and to their natural protein variants or protein variants redesigned by engineering, as well as to peptidomimetic molecules derived from the above and used as antitumor agents.
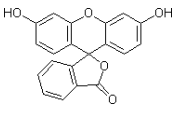
A Kinetic Study on the Electrochemical Bromination of Fluorescein to Eosin in a Batch Reactor
Vasudevan, D.,Basha, C. A.
, p. 165 - 166 (2007/10/03)
The bromination of fluorescein to eosin has been carried out in a batch reactor in NaHCO3 medium using TSI anode, stainless steel cathode and nylon as the separator. The kinetics of the reaction have been followed by uv-visible spectra of samples withdrawn at various time intervals during the electrolysis. Probable mechanism for the bromination of fluorescein has been suggested and attempts have been made for reaction modeling.
Acid Red 87 Upstream and downstream
17372-87-1 Upstream product
17372-87-1 Downstream Products