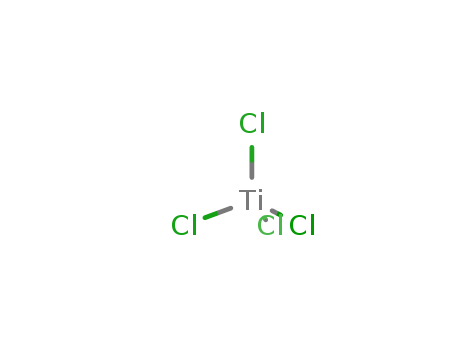
Titanium(IV) chloride literature
Preparation and properties of some new trifluoromethanesulfonates
Noftle, Ronald E.,Cady, George H.
, p. 2182 - 2184 (1966)
Nitrosyl trifluoromethanesulfonate, NOSO3CF3, has been prepared by the reaction of NO with (CF3SO3)2 and quantitatively by the reaction of NOCl with CF3SO3H. Titanium tetrachlori
Heterogeneous Catalysis in Solution. Part 27. - Reaction between Titanium(III) and Triiodide Ions Catalysed by Platinum
Xiao, Shaorong,Spiro, Michael
, p. 1983 - 1986 (1994)
The rate of the reaction between titanium(III) chloride and triiodide ions in an acid chloride medium has been studied in the absence and presence of a rotating platinum disk.At low concentrations of triiodide the catalytic rate at platinum was found to be first order in I3(-), zero order with respect to Ti(III) and H(+), and proportional to the square root of the rotation speed of the disk.These results pointed to diffusion-controlled catalysis, as did the low activation energy of 19 kJ mol-1 as compared with 40 kJ mol-1 for the uncatalysed reaction.All these findings are consistent with an electrochemical interpretation of the catalytic mechanism, as was shown by the current-potential curves determined for the two reactants at the same reduced platinum surface.Provided that the voltammograms for Ti(III) had been carried out in the presence of the same concentration of KI as in the reaction mixtures, the rates and mixture potentials determined electrochemically agreed well with the catalytic rates and potentials measured experimentally.The fact that quite different results were obtained when no KI was present in the TiCl3 solution provides further confirmation for the modified form of the additivity principle.
Impact of synthesis conditions on surface chemistry and structure of carbide-derived carbons
Portet, Cristelle,Kazachkin, Dmitry,Osswald, Sebastian,Gogotsi, Yury,Borguet, Eric
, p. 137 - 142 (2010)
Carbide-derived carbons produced by chlorination of titanium carbide at 600, 800, or 1100 °C were subjected to a post-treatment at 600 °C in Ar, H2, or NH3 atmosphere. Experimental results suggest that the chlorination temperature influences the ordering of carbon in a manner that impacts specific surface area and porosity. Higher chlorination temperatures lead to higher total pore volume and increased ordering, but lower microporosity. The effect of post-treatments on surface chemistry is pronounced only for samples chlorinated at 600 °C; post-treatments in Ar are shown to be less effective for chlorine removal than those performed in H2 or NH3. Post-treatments in Ar result in a lower total pore volume compared to the ones in H2 or NH3 for the same chlorination temperature. Samples chlorinated at higher temperatures contained less oxygen functionalities than samples chlorinated at 600 °C, and showed correspondingly less desorption of H2O, possibly due to diminished uptake of ambient water.
Compatibility of TiO2 with a CaO-CaCl2 melt
Lebedev,Sal'nikov,Tarabaev,Sizikov,Rymkevich
, p. 1491 - 1497 (2008/03/15)
Interaction of TiO2 with a CaO-CaCl2 melt was studied to determine whether electrolytic production of titanium from oxide raw materials is possible.
The low temperature synthesis of metal oxides by novel hydrazine method
Rane,Uskaikar,Pednekar,Mhalsikar
, p. 627 - 638 (2008/10/09)
The hydroxide, oxalate and citrate precursors of the metal oxides such as γ-Fe2O3, (MnZn)Fe2O4, Cu(K)Fe2O4, BaTiO3, La(Sr)MnO3, La(Sr)AlO3, La/Gd(Ca/Ba/Sr)C
Electron deficient carbon-titanium triple bonds: Formation of triplet XC÷TiX3 methylidyne complexes
Lyon, Jonathan T.,Andrews, Lester
, p. 9858 - 9863 (2008/10/09)
Laser-ablated titanium atoms react with CX4 (X = F and Cl) to produce triplet state XC÷TiX3 complexes trapped in an argon matrix. Products are identified by their infrared spectra and comparison to theoretically predicted vibrations.